An experiment in reconstructing primordial proteins solves a long-standing riddle
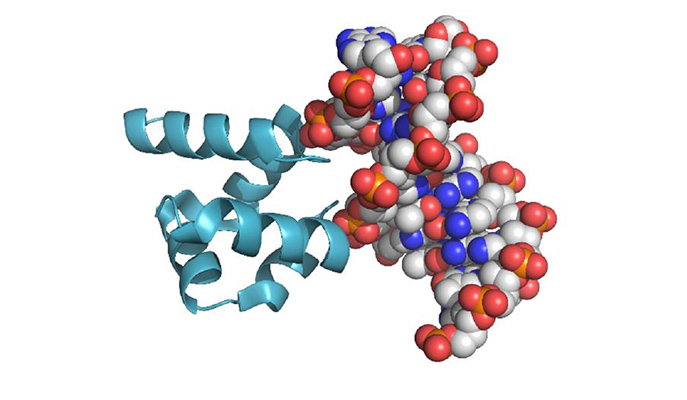
The characteristic (HhH)2 fold and its binding to the minor groove of a modern DNA molecule. How did the first ones form?
What did the very first proteins look like – those that appeared on Earth around 3.7 billion years ago? Prof. Dan Tawfik of the Weizmann Institute of Science and Prof. Norman Metanis of the Hebrew University of Jerusalem have reconstructed protein sequences that may well resemble those long-lost ancestors of modern proteins, and their research suggests a way that these primitive proteins could have played a role in forming the earliest living cells. Their findings were published in the Proceedings of the National Academy of Sciences (PNAS).
The proteins encoded in a cell’s genetic material are the screws, springs and cogs of a living cell – all of its resting and moving parts. But the first proteins, we assume, appeared well before cells and life as we know it. Modern proteins are made of 20 different amino acids, all of them essential to protein-building, and all arranged in the form of a polymer – a long, chain-like molecule – in which the placement of each amino acid is crucial to the protein’s function. But the emergence of the earliest proteins presents a paradox. Because the amino acids needed to make proteins are themselves produced by other proteins – enzymes. It’s a chicken-and-egg kind of question, and it has only been partially answered until now.
One vital type of amino acid has been missing from that experiment and every experiment that followed in its wake
Scientists believe that the very first true proteins materialized from shorter segments called peptides. These peptides would have been assembled from those amino acids that were spontaneously created in the primordial chemical soup; the short peptides would have then bound to one another, over time producing a protein capable of some sort of action. The spontaneous generation of amino acids had already been demonstrated in 1952, in the famous experiment by Miller and Urey, in which they replicated the conditions thought to exist on Earth prior to life, including energy coming from lightning or volcanoes. Showing amino acids could, under the right conditions, form without help from enzymes’ or any other mechanism now seen in a living organism, suggested that amino acids were the “egg” that preceded the enzyme “chicken.”
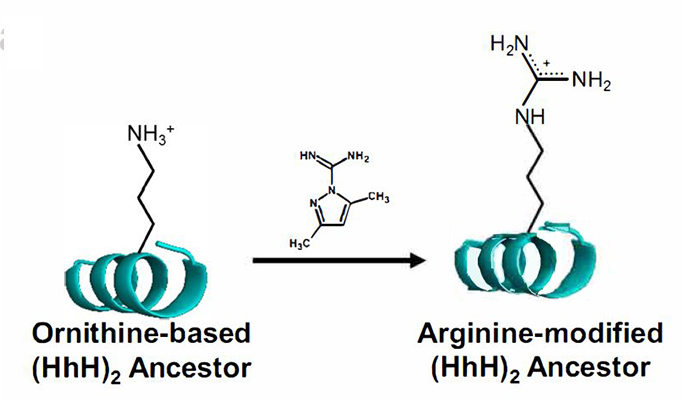
Ornithine to arginine in a simple chemical reaction
Tawfik, who is in the Institute’s Biomolecular Sciences Department, says that this is all well and good, “but one vital type of amino acid has been missing from that experiment and every experiment that followed in its wake: amino acids like arginine and lysine that carry a positive electric charge.” These so-called cationic amino acids are particularly important to modern proteins, as they interact with DNA and RNA, both of which carry net negative charges. RNA is today presumed to be the original molecule that could both carry information and make copies of itself, so contact with positively-charged amino acids is deemed necessary for further steps in the development of living cells to occur.
But there was one positively-charged amino acid that appeared in the Miller-Urey experiments, an amino acid called ornithine that is today found as an intermediate step in arginine production, but is not, itself, used to build proteins. The research team asked: What if ornithine was the positive amino acid in those ancestral proteins? They designed an original experiment to test this hypothesis.
The scientists began with a relatively simple protein from a family that binds to DNA and RNA, applying phylogenetic methods to infer the sequence of the ancestral protein. This protein would have been rich in positive charges – 14 of the 64 amino acids being either arginine or lysine. Next, they created synthetic proteins in which ornithine replaced these as the positive charge carrier.

Proteins with primitive arginine-based proteins (right) might have been capable of self-assembly and phase separation to create cell-like droplets
The ornithine-based proteins bound to DNA, but weakly. In Metanis’ lab, however, the researchers applied simple chemical reactions that could convert ornithine to arginine. And these chemical reactions occurred under those conditions assumed to have prevailed on Earth at the time the first proteins would have appeared. As more and more of the ornithine was converted to arginine, the proteins came more and more to resemble modern proteins, and to bind to DNA in a way that was stronger and more selective.
The scientists also discovered that in the presence of RNA, that the ancient form of the peptide engaged in phase separation (like oil drops in water) – a step that can then lead to self-assembly and compartmentalization. And this, says Tawfik, suggests that such proteins, together with RNA, could form proto-cells, from which living cells as we know them now might have evolved.
Prof. Dan Tawfik is the incumbent of the Nella and Leon Benoziyo Professorial Chair.

Recent Comments