Psychedelics are a hot topic in labs all over the world because they hold great potential for relieving the symptoms of depression, anxiety, PTSD and other mood-related conditions. Still, there is a major hurdle to developing these substances into safe, effective medications: Very little is known about how psychedelic drugs work.
In a study reported recently in Molecular Psychiatry, a team headed by Dr. Takashi Kawashima at the Weizmann Institute of Science has developed a new approach that makes it possible to observe how psychedelics influence behavior and how they affect individual cells in the brain. The method combines powerful optical microscopy, advanced image analysis and artificial intelligence, and uses immature, larval zebrafish as its animal model.

Targeting serotonin for mental health
Psychedelics have been with us for thousands of years, from ancient shamanistic rituals to today’s wild parties. They were placed off limits for scientific study by the Comprehensive Drug Abuse Prevention and Control Act of 1970, which inaugurated the United States’ “war on drugs.” Recently, however, psychedelics have made it to the right side of the law.
According to Kawashima, who in addition to being a research neuroscientist is a medical doctor, psychedelics are now poised to do something that’s truly on the up and up: improve the state-of-the-art for the treatment of mood-related psychiatric conditions. In particular, a number of psychedelics are currently being investigated for their effects on serotonin, a chemical that, among its many other functions, carries messages throughout the brain and nervous system, regulating mood.
“Psychedelics affect serotonin receptors much faster than common antidepressants and appear to act in a more targeted manner”
Kawashima points out, however, that it’s problematic to test psychedelics on humans because of their hallucinogenic side effects, and it’s hard to know what exactly they do to the brain because they may target circuits in the brain’s deep-seated regions, where neural activity is difficult to observe. “Zebrafish larvae, on the other hand, are transparent, making it possible to monitor drugs’ impact on specific brain cells and to correlate this with behavior.”
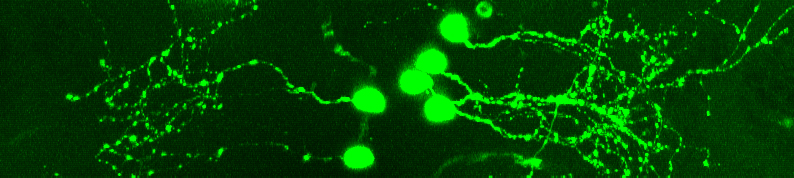
The present study was launched at the instigation of Dr. Dotan Braun, a psychiatrist who joined Kawashima’s lab in Weizmann’s Brain Sciences Department as a visiting scientist. Inspired by Kawashima’s technology for imaging brain activity in zebrafish and his investigations of the serotonin system, Braun proposed a project that would help clarify the precise effects of psychedelics on serotonin. This in turn might contribute to the development of potential psychedelic alternatives to the widely prescribed class of antidepressants known as serotonin-selective reuptake inhibitors, or SSRIs, which include such drugs as Cipralex and Prozac.
“SSRIs elevate serotonin levels throughout the brain,” Braun says. “Psychedelics, in contrast, affect serotonin receptors via a different, much faster mechanism and they appear to act on brain areas in a more targeted manner. A better understanding of their mechanism of action and a mapping of their influence on the brain may lead to more efficient drugs, with fewer side effects.”
Fish on drugs
The scientists designed an experiment that enabled them to “get into the head” of zebrafish soaked in a solution containing psilocybin, a mushroom-derived psychedelic compound being tested for use against depression that is not relieved by other medications. Following a four-hour psilocybin “bath,” the fish dove into Kawashima’s arena for behavioral experiments: a shallow pool of water that has attention-grabbing visual patterns projected onto its glass bottom.
After exposing the fish to a stressful situation – a sudden, temporary drop in water temperature – the researchers compared their behaviors to those of fish that had not taken a preparatory bath. “We wanted to see how psychedelics affect the fish’s stress response,” Kawashima says, adding with a smile: “We found that, similar to what can be true for humans, when you’re heading into a stressful situation, taking a long bath can help.”

Indeed, the psychedelic bath reduced stress-related behaviors in two ways. Following stress exposure, the presoaked fish were more likely to explore the tank, venturing even into its darker domains, compared to fish that were drug-free. The “drugged” fish also darted about faster than the “sober” ones. These differences suggested that psilocybin produced a stimulating effect.
In addition, psilocybin reduced post-stress anxiety. “Fish that had not bathed in psilocybin reacted to the sudden drop in temperature with irregular, zig-zag swimming,” says Ayelet Rosenberg, a research student in Kawashima’s lab who, together with Braun, is a first coauthor of the research paper. “But the fish that had been pretreated with the psychedelic drug stayed calm; they seemed to take this added stress in stride.”
The scientists were able to pick up on these differences in behavior by documenting freely swimming individual zebrafish in minute detail with the help of a high-speed camera that produced 270,000 images for each 15-minute experiment. A subset of this wealth of images was manually annotated for ten zebrafish body parts, including the eyes, nostril, body trunk, and six points along the tail, and used to train a deep neural network – an advanced AI algorithm – to identify the nuances of the fish’s swimming patterns. Once trained, the algorithm was able to identify complex swimming trajectories and map out how behavior changed in fish under the influence of psychedelics.
The scientists were then able to link these behavioral effects to specific neural activation patterns. They relied on a method, previously developed by Kawashima and colleagues, involving the fluorescent tagging of individual zebrafish neurons and neural circuits, which causes them to light up when activated. Because the zebrafish larvae are transparent, the scientists were able to use a powerful optical microscope to image this activation directly, enabling them to identify specific changes in serotonin-related neurons and circuits.
Over 100: number of visits, phone calls and emails to Israel’s Ministry of Health by members of Dr. Kawashima’s team before they received permission to use psilocybin, making theirs the first Israeli lab ever authorized to work with this controlled psychedelic substance.
“Our optical imaging has revealed neural activity patterns in psilocybin-soaked fish that were similar to those seen by other labs in the mammalian brain exposed to psychedelics,” says Kawashima. “This indicates that psilocybin exerts its influence on behavior through neural mechanisms in deep areas of the brain that have been conserved in evolution and are also found in mammals, including humans.”
A “trip” toward better psychiatric treatment
The Kawashima team’s methodology and findings could help advance the development of psychedelics as therapies for mood-related conditions. Kawashima cautions that studying psychedelics in fish has its limits: Despite the fascinating nature of the question, it is unclear, for example, whether zebrafish experience hallucinatory “trips” in the course of these investigations. Still, his method can help advance therapeutic research in psychiatry.
“The practical significance of our work is that it demonstrates a fish-based screening tool for drug discovery,” he says. “Researchers can use our method to test new drug compounds or compare the relative usefulness of the serotonin-targeting drugs already in use. This could lead to discoveries about the mechanics of serotonin-related disorders, something that could generate entirely new approaches to the treatment of depression, anxiety, OCD, PTSD and addiction.”
Dr. Takashi Kawashima is the incumbent of the Birnbach Family Career Development Chair.
His research is supported by the Swiss Society Center for Research on Perception and Action and the Jared M. Drescher Center for Research on Mental and Emotional Health.

Recent Comments